main content It’s a Small World
-
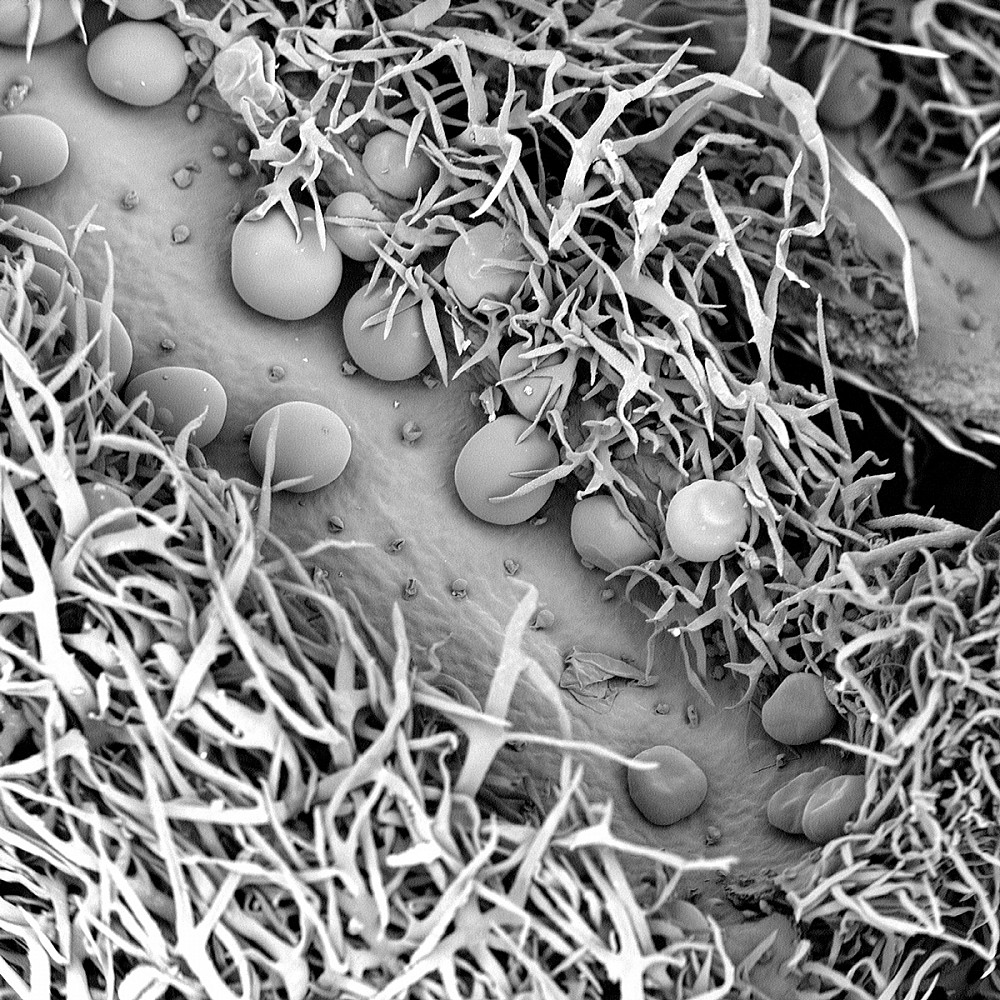
 LAVENDER BUD | This image shows the bud’s branch-like structures (nonglandular trichomes), which help protect it from the elements and insects, and its spherical structures (glandular trichomes), which hold the plant’s oils. Anna DePaepe BA ’19 says, “The classical lavender scent is released when the glandular trichomes flatten out and release their oils, similar to how a balloon deflates.” (Captured by Anna DePaepe BA ’19; Major: Biochemistry and Molecular Biology; Magnification 290x).
LAVENDER BUD | This image shows the bud’s branch-like structures (nonglandular trichomes), which help protect it from the elements and insects, and its spherical structures (glandular trichomes), which hold the plant’s oils. Anna DePaepe BA ’19 says, “The classical lavender scent is released when the glandular trichomes flatten out and release their oils, similar to how a balloon deflates.” (Captured by Anna DePaepe BA ’19; Major: Biochemistry and Molecular Biology; Magnification 290x). -
 ELEPHANT STAG BEETLE ANTENNA | “I love to show people that insects are really quite gorgeous if you look at them through a different lens,” says Ryan Cook BA ’19. Using a beetle that he collected from his home state of North Carolina, Ryan imaged the insect’s sensilla. These hair-like structures function much like our noses or tongues and help the beetle detect everything from rotting fruit to a possible mate. (Captured by Ryan Cook BA ’19; Major: Biology; Magnification 325x).
ELEPHANT STAG BEETLE ANTENNA | “I love to show people that insects are really quite gorgeous if you look at them through a different lens,” says Ryan Cook BA ’19. Using a beetle that he collected from his home state of North Carolina, Ryan imaged the insect’s sensilla. These hair-like structures function much like our noses or tongues and help the beetle detect everything from rotting fruit to a possible mate. (Captured by Ryan Cook BA ’19; Major: Biology; Magnification 325x). -
 FERN FROND | “As one of the oldest groups of plants, with fossils dating back over 300 million years ago, I thought if any plant deserved some close attention it should be a fern,” says Emma Wood BA ’19. This image shows the deflated spore capsules on the underside of a frond from a diamondleaf fern, a plant that grows in abundance around the Frank Manor House. (Captured by Emma Wood BA ’19; Major: East Asian Studies; Magnification 490x).
FERN FROND | “As one of the oldest groups of plants, with fossils dating back over 300 million years ago, I thought if any plant deserved some close attention it should be a fern,” says Emma Wood BA ’19. This image shows the deflated spore capsules on the underside of a frond from a diamondleaf fern, a plant that grows in abundance around the Frank Manor House. (Captured by Emma Wood BA ’19; Major: East Asian Studies; Magnification 490x). -
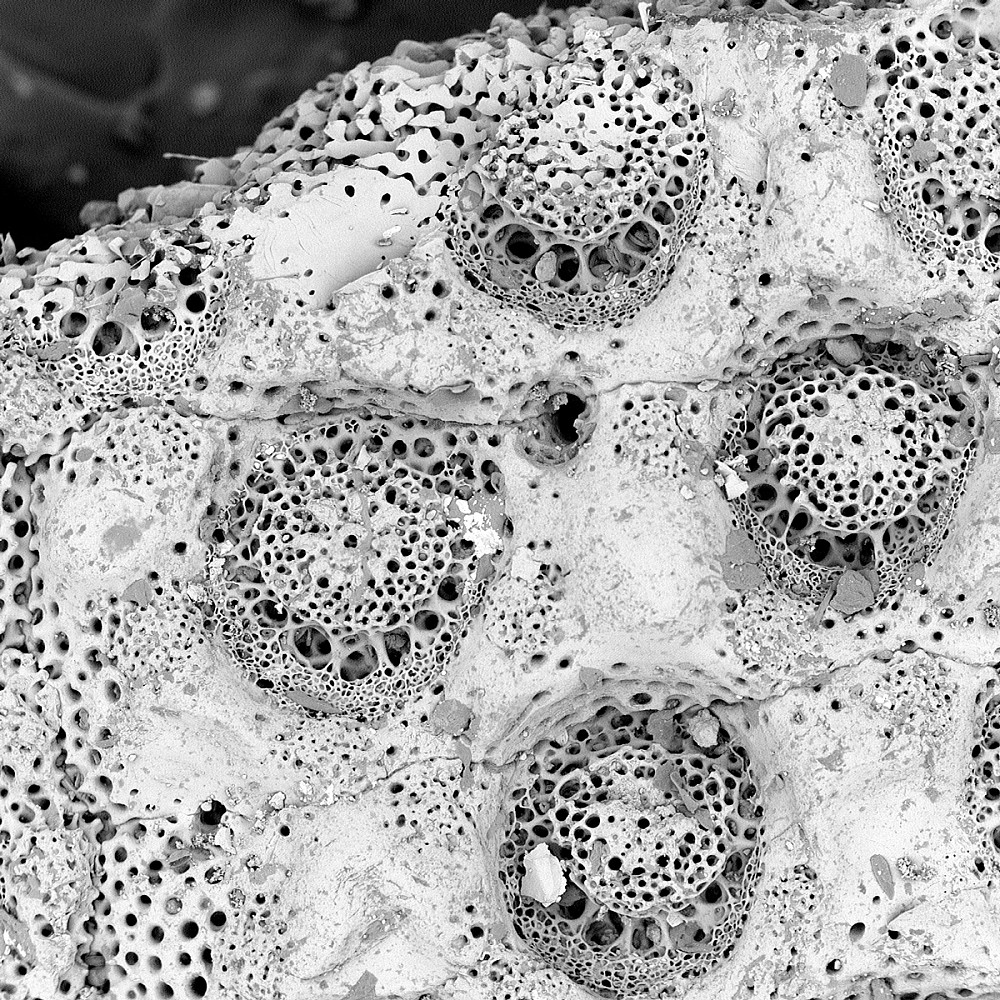
 SAND DOLLAR | A native of Santa Barbara, California, Aria Ballance BA ’19 brought several sand dollars with her to college to remind her of home and bring good luck. This image shows part of the petal-like pattern on top of the sand dollar. The cheese-like holes indicate the hair-like cilia that grew out of the sand dollar when it was alive. (Captured by Aria Ballance BA ’19; Major: Chemistry; Magnification 320x).
SAND DOLLAR | A native of Santa Barbara, California, Aria Ballance BA ’19 brought several sand dollars with her to college to remind her of home and bring good luck. This image shows part of the petal-like pattern on top of the sand dollar. The cheese-like holes indicate the hair-like cilia that grew out of the sand dollar when it was alive. (Captured by Aria Ballance BA ’19; Major: Chemistry; Magnification 320x). -
 TABLE SALT | Sodium chloride contains striking cracks and cubical structures when magnified. “It looks as though someone took a chisel to it,” says Frankie Warden BA ’20. She attributes this to the underlying molecular structure of salt—it’s similar to how concrete blocks line up to build a wall, complete with cracks in the mortar. (Captured by Frankie Warden BA ’20; Major: Chemistry; Magnification 785x).
TABLE SALT | Sodium chloride contains striking cracks and cubical structures when magnified. “It looks as though someone took a chisel to it,” says Frankie Warden BA ’20. She attributes this to the underlying molecular structure of salt—it’s similar to how concrete blocks line up to build a wall, complete with cracks in the mortar. (Captured by Frankie Warden BA ’20; Major: Chemistry; Magnification 785x). -
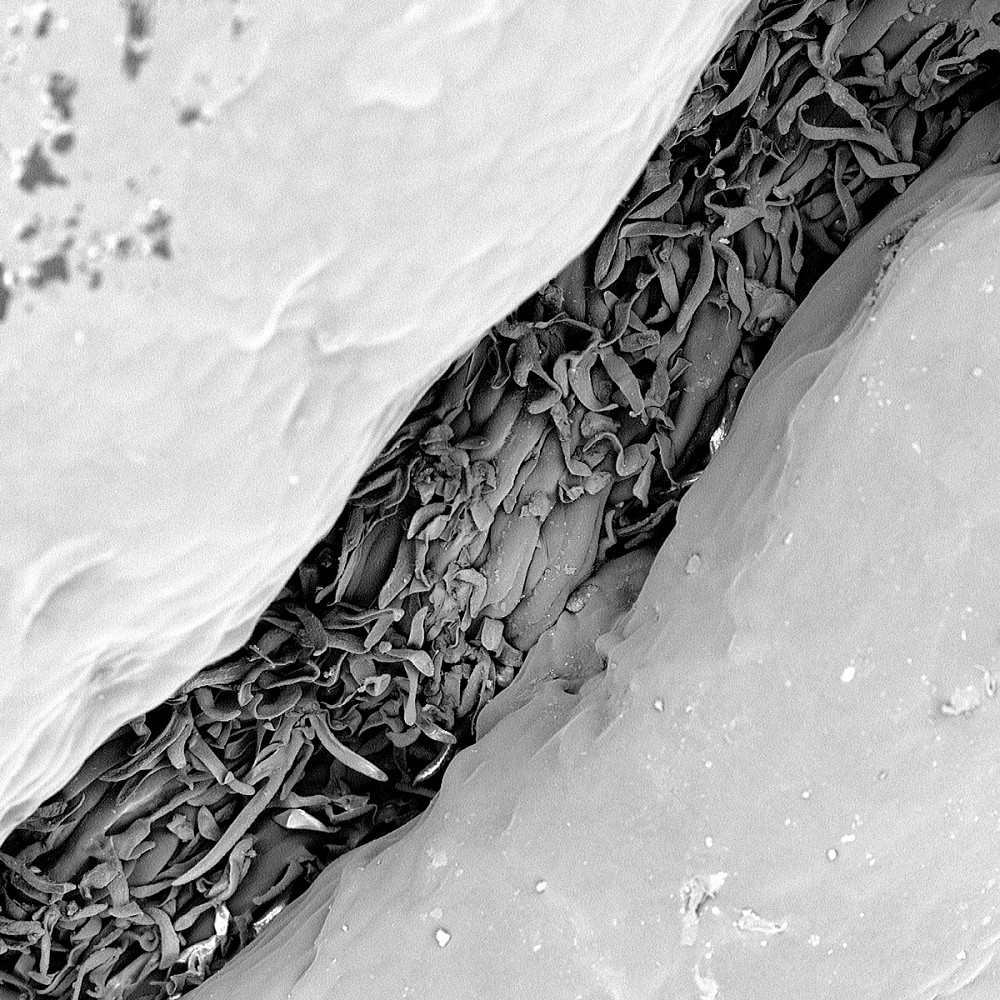
 CHAMOIS LEATHER | A common artist’s tool, chamois leather is frequently preferred over fingers for blending charcoal. If Cayden Bullock BA ’19 were not a dedicated chemist, he would be an illustrator. “I grew up connecting with my father through comic books,” he says. “I like to involve art whenever I can.” Here, his two passions merged as he illuminated the superior blending properties of the leather’s fibrous surface. (Captured by Cayden Bullock BA ’19; Major: Chemistry; Magnification 320x).
CHAMOIS LEATHER | A common artist’s tool, chamois leather is frequently preferred over fingers for blending charcoal. If Cayden Bullock BA ’19 were not a dedicated chemist, he would be an illustrator. “I grew up connecting with my father through comic books,” he says. “I like to involve art whenever I can.” Here, his two passions merged as he illuminated the superior blending properties of the leather’s fibrous surface. (Captured by Cayden Bullock BA ’19; Major: Chemistry; Magnification 320x). -
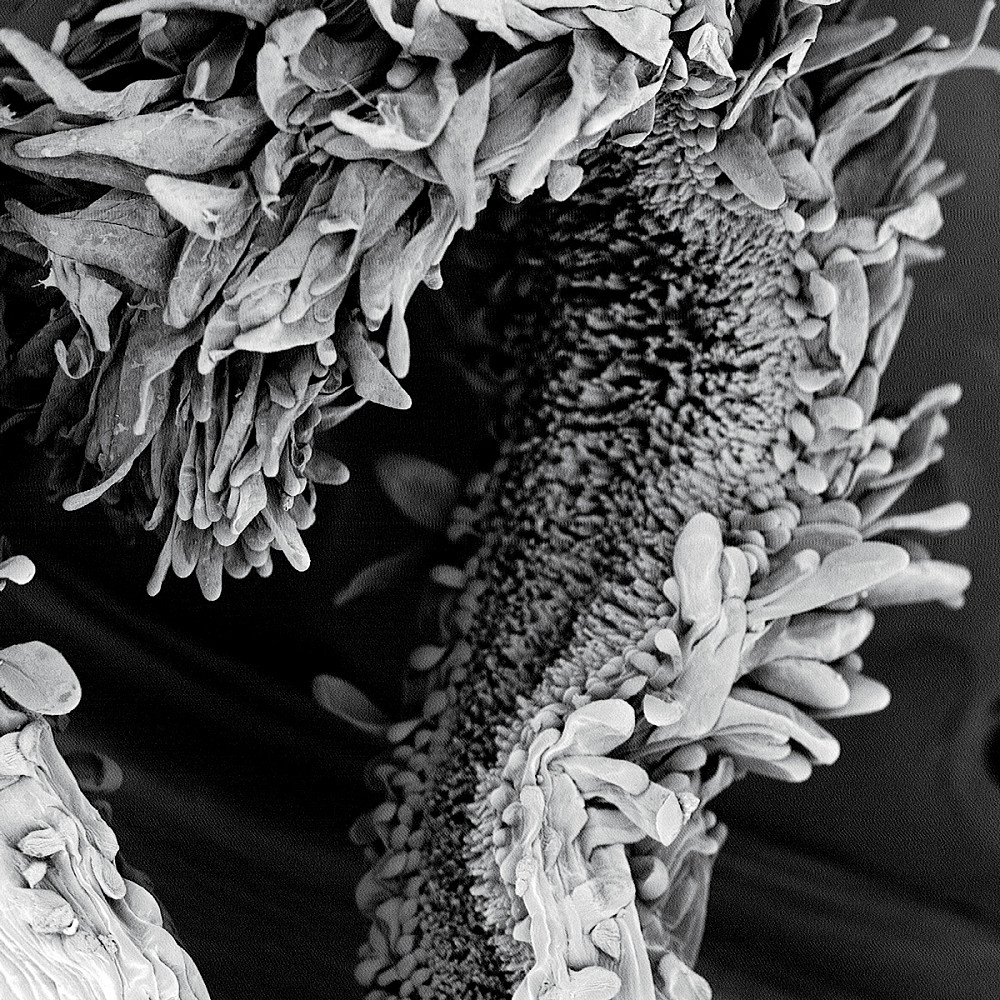
 SUNFLOWER | “Named for its radiant appearance, the common sunflower is composed of thousands of smaller flowers budding outwards from its round head,” says Sally Chaffee BA ’19. The flowers at the dark center include the male and female reproductive organs. This image shows a single female stigma and the varied protruding hairs that make it so successful in capturing male-produced pollen. (Captured by Sally Chaffee BA ’19; Major: Chemistry; Magnification 330x).
SUNFLOWER | “Named for its radiant appearance, the common sunflower is composed of thousands of smaller flowers budding outwards from its round head,” says Sally Chaffee BA ’19. The flowers at the dark center include the male and female reproductive organs. This image shows a single female stigma and the varied protruding hairs that make it so successful in capturing male-produced pollen. (Captured by Sally Chaffee BA ’19; Major: Chemistry; Magnification 330x). -
 BANDAGE | This image shows the nonadhesive plastic side of a common bandage. It’s made of woven polyethylene, a strong, flexible fabric that appears as an equally spaced grid pattern under magnification. Kindra Jones BA ’19 says, “It was a challenge to narrow down all of the beautiful images to just one.” (Captured by Kindra Jones BA ’19; Major: Chemistry; Magnification 350x).
BANDAGE | This image shows the nonadhesive plastic side of a common bandage. It’s made of woven polyethylene, a strong, flexible fabric that appears as an equally spaced grid pattern under magnification. Kindra Jones BA ’19 says, “It was a challenge to narrow down all of the beautiful images to just one.” (Captured by Kindra Jones BA ’19; Major: Chemistry; Magnification 350x). -
 ROSEMARY LEAF | For Keldy Mason BA ’20, rosemary represents an “academic scent” since it grew near both his middle school and his high school. In fact, he used to fill his locker with dried rosemary because he liked its aroma. This image shows the leaf’s waxy top and hairy underside. The leaf’s hairs hold the oil that produces rosemary’s unique scent and taste. (Captured by Keldy Mason BA ’20; Major: Chemistry; Magnification 420x).
ROSEMARY LEAF | For Keldy Mason BA ’20, rosemary represents an “academic scent” since it grew near both his middle school and his high school. In fact, he used to fill his locker with dried rosemary because he liked its aroma. This image shows the leaf’s waxy top and hairy underside. The leaf’s hairs hold the oil that produces rosemary’s unique scent and taste. (Captured by Keldy Mason BA ’20; Major: Chemistry; Magnification 420x). -
 IBUPROFEN | Nat Richman BA ’19, an accomplished pitcher on L&C’s baseball team, uses ibuprofen to relieve the aches and pains of his sport. His image captures a clump of ibuprofen crystal growth that displays a lack of geometric pattern. If he were to continue this work, he would research the possibility of creating a structure with “increased geometric conformity,” which he predicts would increase the solubility and effectiveness of the drug. (Captured by Nat Richman BA ’19; Major: Chemistry; Magnification 2460x).
IBUPROFEN | Nat Richman BA ’19, an accomplished pitcher on L&C’s baseball team, uses ibuprofen to relieve the aches and pains of his sport. His image captures a clump of ibuprofen crystal growth that displays a lack of geometric pattern. If he were to continue this work, he would research the possibility of creating a structure with “increased geometric conformity,” which he predicts would increase the solubility and effectiveness of the drug. (Captured by Nat Richman BA ’19; Major: Chemistry; Magnification 2460x).
L&C Magazine is located in McAfee on the Undergraduate Campus.
MSC: 19
email magazine@lclark.edu
voice 503-768-7970
fax 503-768-7969
The L&C Magazine staff welcomes letters and emails from readers about topics covered in the magazine. Correspondence must include your name and location and may be edited.
L&C Magazine
Lewis & Clark
615 S. Palatine Hill Road
Portland OR 97219